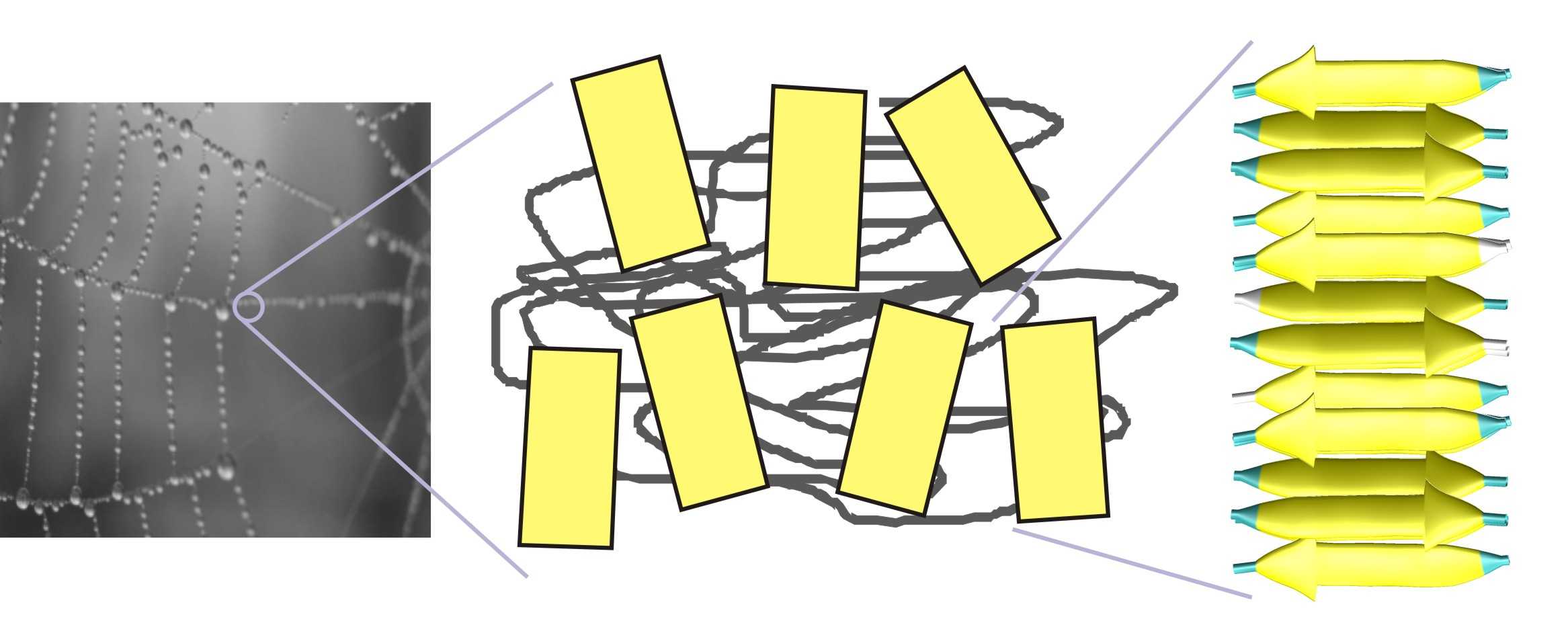
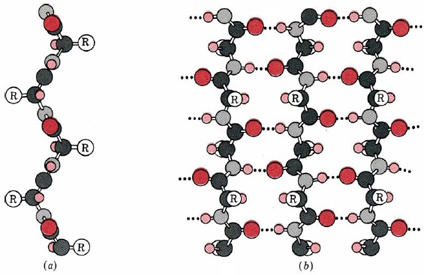
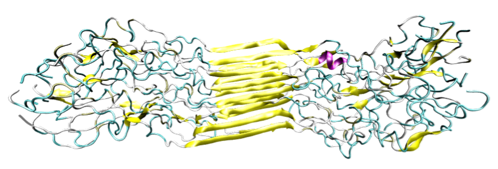
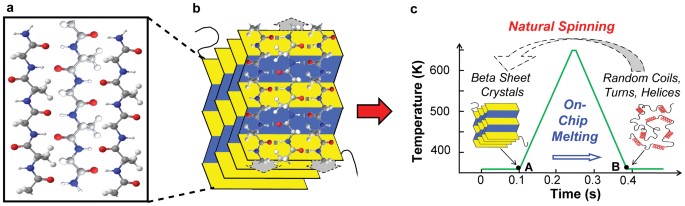
![PDF] Molecular and nanostructural mechanisms of deformation, strength and toughness of spider silk fibrils. | Semantic Scholar PDF] Molecular and nanostructural mechanisms of deformation, strength and toughness of spider silk fibrils. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/daa6b91a8a661077da1f12c2de436e945172c41e/17-Figure1-1.png)
PDF] Molecular and nanostructural mechanisms of deformation, strength and toughness of spider silk fibrils. | Semantic Scholar
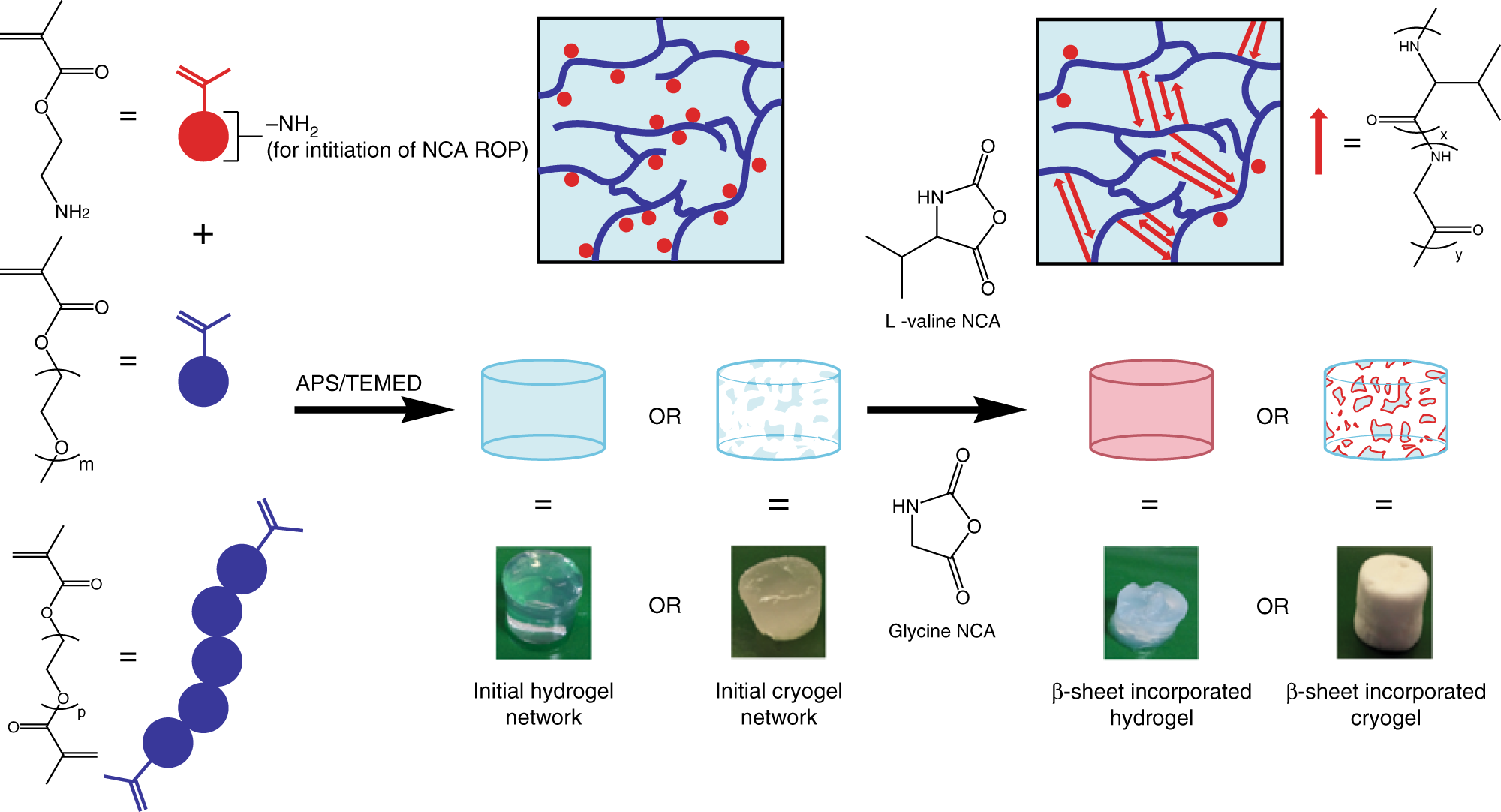
Spider-silk inspired polymeric networks by harnessing the mechanical potential of β-sheets through network guided assembly | Nature Communications

Influence of Water Content on the β-Sheet Formation, Thermal Stability, Water Removal, and Mechanical Properties of Silk Materials | Biomacromolecules

Dissecting the structural determinants for the difference in mechanical stability of silk and amyloid beta-sheet stacks - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C3CP00067B
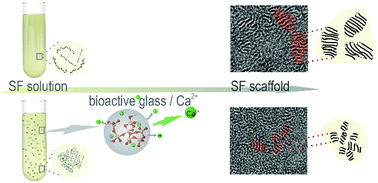
Size of silk fibroin β-sheet domains affected by Ca2+ - Journal of Materials Chemistry B (RSC Publishing)

Optimal Protein Sequence Design Mitigates Mechanical Failure in Silk β-Sheet Nanocrystals | ACS Biomaterials Science & Engineering

Optimal Protein Sequence Design Mitigates Mechanical Failure in Silk β-Sheet Nanocrystals | ACS Biomaterials Science & Engineering
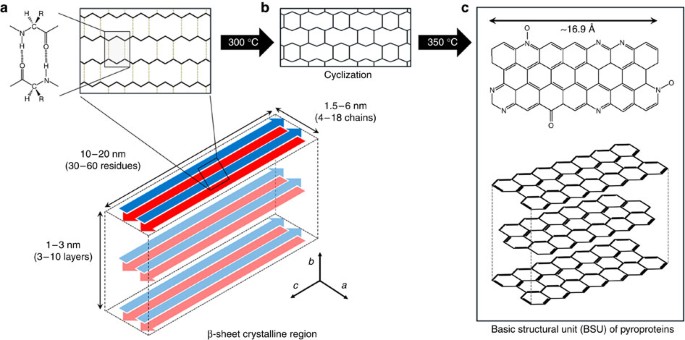
Carbonization of a stable β-sheet-rich silk protein into a pseudographitic pyroprotein | Nature Communications