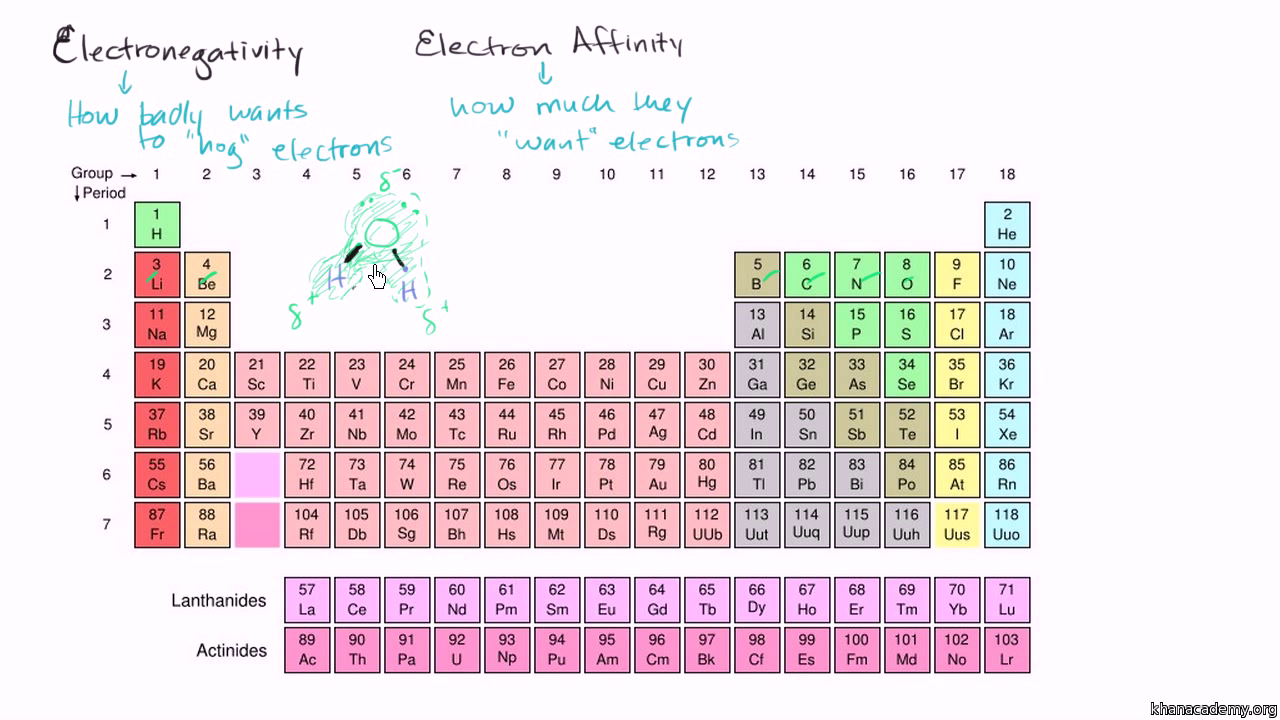
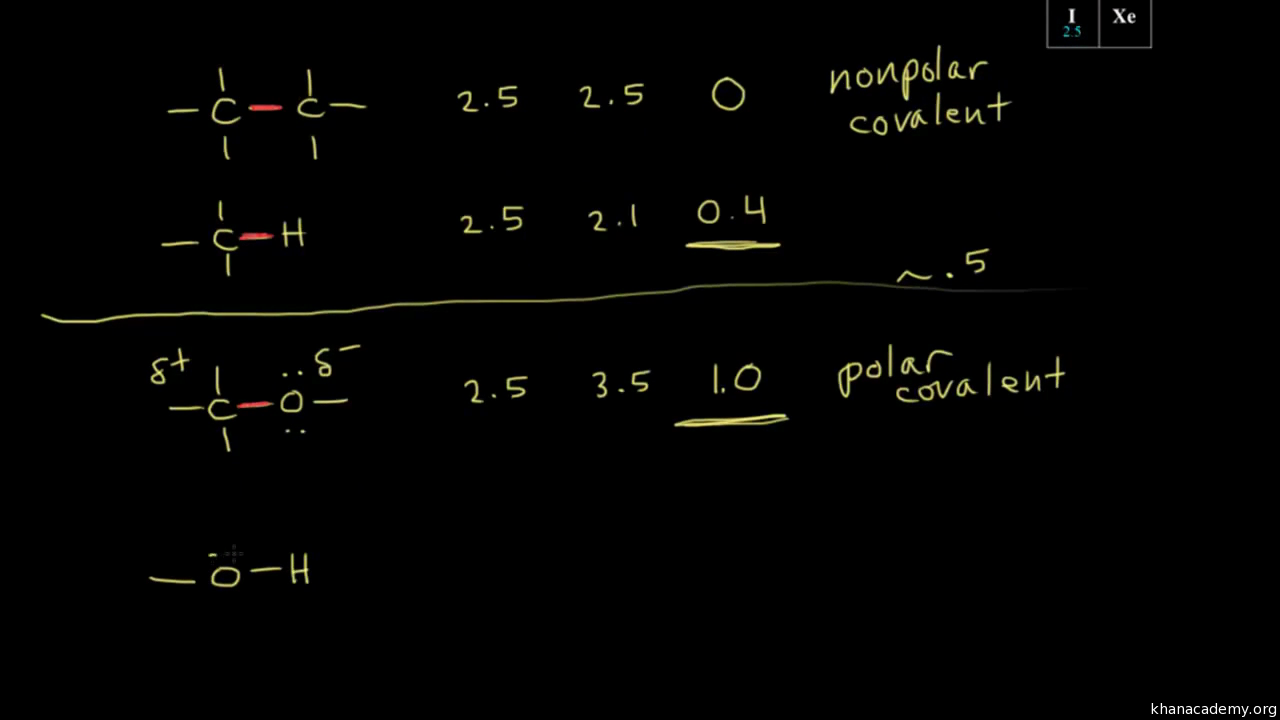Electronegativity, Basic Introduction, Periodic Trends - Which Element Is More Electronegative? - YouTube

ilustration Calculate the electronegativity of carbon from the following data: E = 104.2 kcal mol-1, Ecc = 83.1 kcal mol-1 Ech = 98.8 kcal mol-1, XH = 2.1 Solution Let the electronegativity

Covalent radius of an element having 82 electrons in extranuclear part and 82 protons in the nucleus is 146 Å. Calculate the electronegativity on Allred Rochow scale of that element.

Calculate the electronegativity value of chlorine on Mulliken's scale, given that IP = 13.0 eV and EA = 4.0 eV.